High quality products to support Pathologists and Biological and Environmental Scientists
Bacteroides (Fecal Contamination) qPCR detection Kit
$376.25This kit is sufficient for 150 reactions:
- 170ul HF183 region specific primer pairs (150 reactions)
- 1.5ml PCR clean water
B. mallei qPCR detection Kit
$376.25This kit is sufficient for 150 reactions:
- 170ul fliP region specific primer pairs (150 reactions)
- 170ul fliP region specific fluorescent probe (150 reactions)
- 1.5ml PCR clean water
B. pseudomallei qPCR detection Kit
$376.25This kit is sufficient for 150 reactions:
- 170ul S664 region specific primer pairs (150 reactions)
- 170ul S664 region specific fluorescent probe (150 reactions)
- 1.5ml PCR clean water
Universal Nucleic Acid ELISA Kit
$399.00- Anti FAM/FITC (01 kits) or Anti Dig (02 kit) Coated 96 well ELISA plate
- Nucleic Acid ELISA Wash Buffer
- Streptavidin HRP
- HRP Substrate (TMB)
- Stop Solution
- Nucleic Acid Calibration Standards for absolute quantification
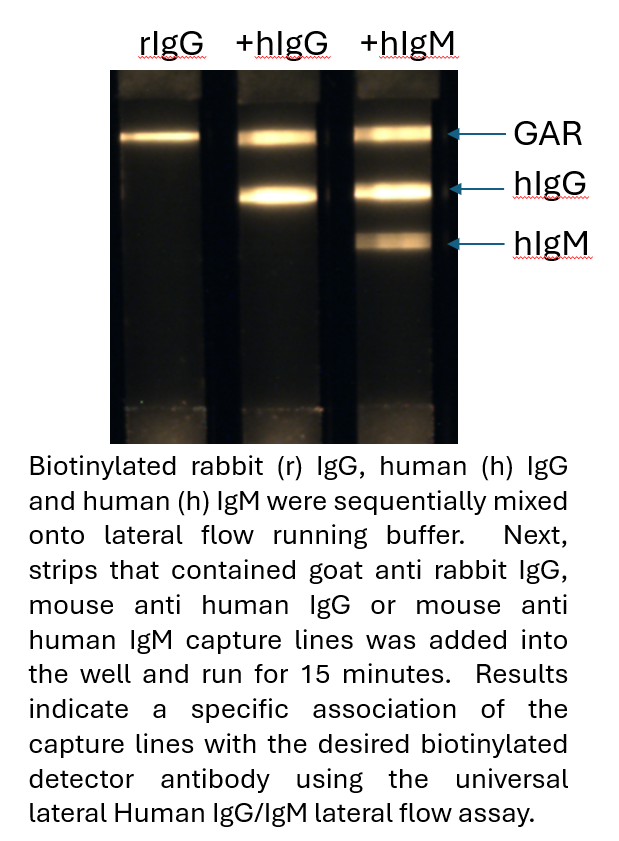
Fluorescent Universal Human IgG/IgM Lateral Flow Serology Kit (Europium)
$322.50Antibody tests are a method of choice to determine if a person has been exposed to a pathogen or not. They are also incredibly valuable in the detection of autoantibodies that can be found in human autoimmune disorders. In this test, a biotinylated antigen (User supplied) is mixed with a biotinylated rabbit IgG (bind to goat anti rabbit control line) and sample (human sera or plasma) is simply mixed into with the specially designed assay running buffer in a well of the supplied 96-well plate, mixed and is then added to the sample port of the cassette. Generally, the reaction is complete in 10-15 minutes. It is very important to note that the relative stoichiometry between the biotinylated antigen, biotinylated rabbit IgG added, and the streptavidin gold is critical for assay optimization. The appropriate concentration of biotinylated antigen to use with strips is dependent upon the purity and sequence and a standard curve can be used to determine the relative ratio (generally between 1ng-100ng per test). A positive control line (biotin-rabbit IgG) antibody will bind to the goat anti rabbit (GAR) line on the test to ensure the assay is running appropriately.
Oligonucleotide-BSA Conjugation Kit
$322.50- Attogene’s Oligonucleotide-BSA Conjugation Kit provides the user with reagents for coupling oligonucleotides to BSA protein.
- Activated BSA (NHS-BSA) supplied in single use vials for convenient coupling of amine-containing DNA or RNA oligonucleotides.
- Simple 90 minute procedure.
- No purification steps required.
- Coupled conjugate ready to use for lateral flow or ELISA-type applications.
- Made by Attogene in the USA