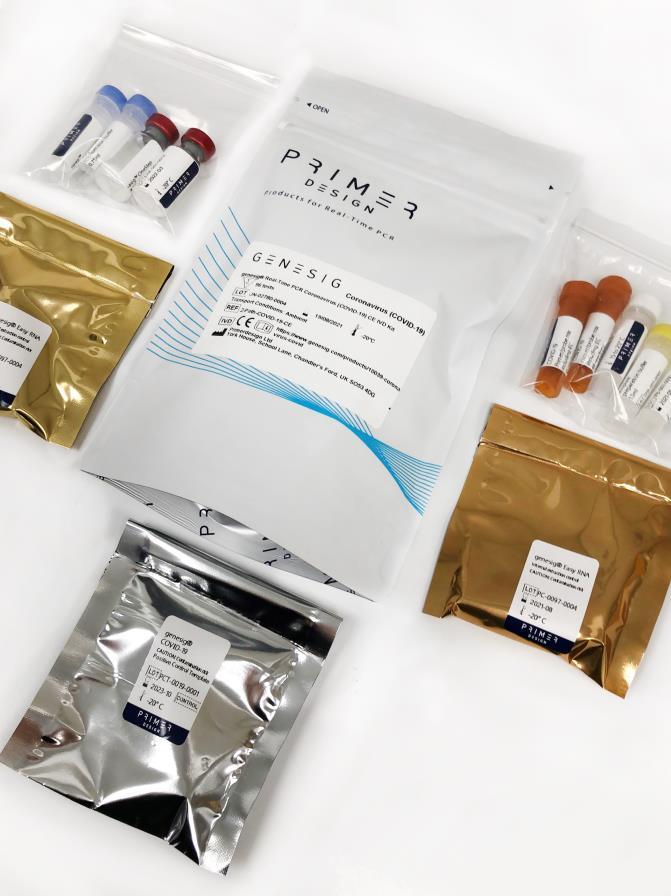
COVID-19 Real-Time PCR assay EUA (USA)
$1,321.04
In Stock & Ready to Ship
Product Features
- • FOR US ONLY
- • Rapid detection and exclusive to the COVID-19 strain
- • Does not detect other related coronavirus strains
- • High priming efficiency
- • Accurate controls to confirm extraction, and assay validity
- • Lyophilised components for ambient shipping
- • Highly specific detection profile
This test has been authorized by FDA under an EUA for use by authorized laboratories.

The Primerdesign Ltd COVID-19 genesig® Real-Time PCR assay is a real-time RT-PCR assay intended for the qualitative detection of nucleic acid from SARS-CoV-2 in oropharyngeal swab specimens from patients suspected of COVID-19 by their healthcare provider. Testing is limited to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. §263a, to perform high complexity tests.
*FDA clinical study. See Instructions for Use for additional clinical data.
You may also like…
oasig Lyophilised OneStep RT-qPCR Master Mix
$277.10150 reaction pack of freeze dried oasig Lyophilised OneStep RT-qPCR Mastermix. 3 x 50 reaction vials and resuspension buffer.
Kit contents
• 3 x lyophilised OneStep Master Mix (50 reactions per glass ampule)
• 1 x lyophilised ROX (BROWN)
• 4 x Resuspension buffer (BLUE)